Studying ADCC & CDC together provides a comprehensive understanding of immune-mediated cytotoxic mechanisms.
ADCC
We investigate antibody-dependent cell-mediated cytotoxicity in whole blood, ensuring preserved native cellular responses.
CDC
Capture the complexity of complement-mediated cytotoxicity and get a holistic view of immune response and therapeutic efficacy.
Physiological Relevance
Our ADCC & CDC assay captures the complete immune response, including interactions between immune cells, complement proteins, cascade systems, and cytokines.
ASSAYS
Why choose ADCC & CDC assays based on whole blood?
- Understand and optimize the risk-benefit ratio of engineered antibodies and antibody-drug conjugates.
- Human whole blood preserves complex interactions between complement, immune cells, cytokines and other blood components.
- Concurrent evaluation of ADCC and CDC, ensuring a physiologically relevant analysis.
- Additional readouts to capture the full immune response.
Simultaneous Assessment of ADCC and CDC in Whole Blood:
- ADCC (Antibody-Dependent Cell-Mediated Cytotoxicity): Evaluated by detecting NK cell activation and target cell depletion.
- CDC (Complement-Dependent Cytotoxicity): Assessed through complement activation and target cell depletion.
- Target Cell Viability: Measured using flow cytometry and hematology parameters to provide insight into cell health and immune interactions.
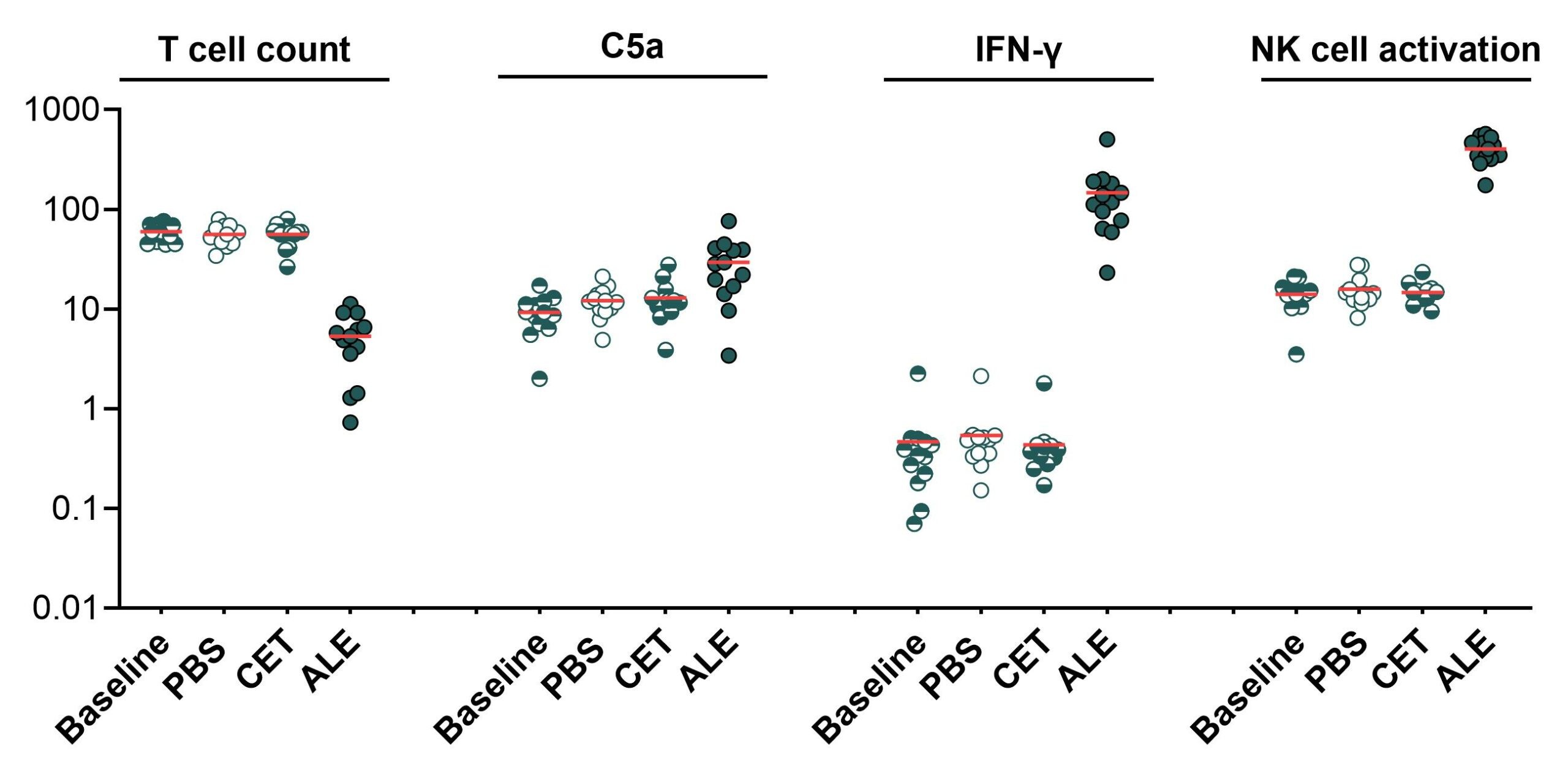
The graphs show a multiparameter analysis of ID.Flow whole blood samples at baseline and after stimulation with vehicle control (PBS), aEGFR (CET), and aCD52 (ALE). CD52 is expressed on many white blood cells such as T cells, whereas EGFR is not expressed in blood. The readouts shown are T cell count , complement activation (C5a release), cytokine production (IFN-γ release), and NK cell activation (CD69 expression).
In addition to ADCC and CDC, ID.Flow can simultaneously assess:
This integrated approach delivers a holistic view of the human immune response ensuring a thorough understanding of an antibody’s impact on the immune responses.
MORE SERVICES

White Blood Cells, Activation & Depletion
Activation of B cells, T cells, NK cells, monocytes, and granulocytes is simultaneously analyzed in fresh human whole blood.
Complement Assays
Assessment of C3a, C5a, and Bb split product in fresh human whole blood with active complement.
Cytokine Release
Sensitive cytokine release prediction in fresh human whole blood.
Platelets, Count & Activation
Platelet activation, conjugates, aggregation, counts, inhibition...